CAPA for Nonconformities
A thorough exploration of Corrective Actions and Preventive Actions (CAPA) as they apply to quality management in GxP-regulated industries. This module covers the identification, categorization, and response to nonconformities, and how CAPA systems ensure continuous quality improvement.
Whether for onboarding or annual refresher training, this Module is seamlessly deployable on any LMS and can be tailored to your company’s exact needs.
Duration: 30 Mins
Grade: Premium Strategic
Description
- Gain a Clear Understanding of Nonconformities and CAPA: Learn to identify and categorize nonconformities and understand how CAPA systems function to prevent recurrence and maintain product quality.
- Master Techniques for Effective CAPA Planning: Discover the elements of a SMART CAPA plan and how to implement actions that are Specific, Measurable, Achievable, Realistic, and Time-bound to address quality issues.
- Enhance Problem-Solving Skills: Distinguish between corrections, corrective actions, and preventive actions, and apply these concepts to real-world quality scenarios within a structured CAPA system.
- Develop Skills in CAPA Effectiveness Verification: Learn how to assess the success of corrective actions and preventive measures over time, ensuring continuous quality improvement and regulatory compliance.
- Apply Practical Knowledge Through Scenario-Based Exercises: Benefit from scenario-based learning and knowledge checks, reinforcing your understanding of CAPA principles and their application to quality management challenges in the life sciences.
Learning Objectives
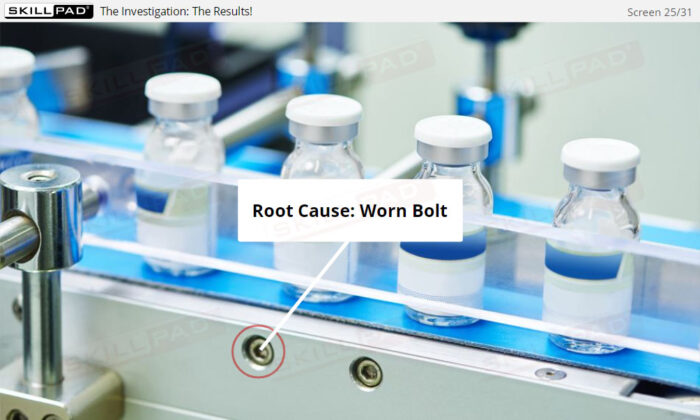
- Explain the term ‘nonconformity’.
- Define CAPA
- Explain the purpose of a CAPA system.
- Explain the difference between minor, major, and critical nonconformities.
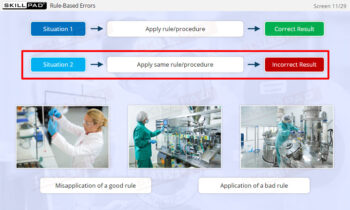
- Distinguish between corrections, corrective actions, and preventive actions.
- Describe the different elements of a ‘SMART ‘CAPA plan.
- Describe the correct way of verifying the effectiveness of a CAPA.
Keywords
- CAPA
- Corrective Actions
- GMP Inspection
- Compliance
- Nonconformities
- Preventive Actions
- Quality Assurance
- Quality System
- Regulatory Compliance
- Risk Management
- SMART CAPA Plan.
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen