GCP Inspection Readiness – Initiate
An overview of a project management approach to preparation for a GCP regulatory inspection, with a focus on the key tasks, roles, and responsibilities of the Initiate phase. Explore how validation of inspection notifications, role assignment, and risk management are essential to a successful inspection outcome.
Whether for onboarding or annual refresher training, this Module is seamlessly deployable on any LMS and can be tailored to your company’s exact needs.
Duration: 30 Mins
Grade: Premium Performance
Description
- Gain a Clear Understanding of Project Management Phases: Learn about the four key phases of an inspection readiness project and the importance of each, with an in-depth focus on the Initiate phase.
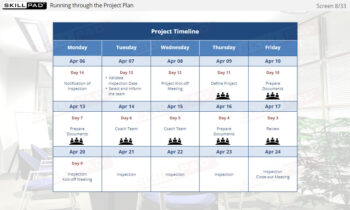
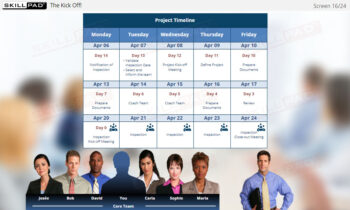
- Perform Key Tasks in the Initiate Phase: Develop practical knowledge of essential tasks, such as confirming inspection notifications, identifying key personnel, and organizing the inspection project kick-off to ensure thorough preparation.
- Improve Risk Management Awareness: Recognize common project management challenges and explore strategies to prevent potential pitfalls during the inspection readiness process.
- Enhance Team Coordination and Role Definition: Learn to establish clear roles and responsibilities, ensuring each team member is prepared to meet inspector expectations and minimize inspection risks.
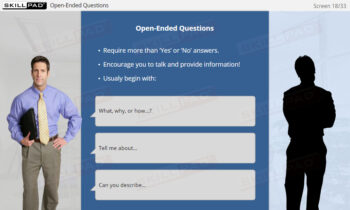
- Apply Project Management Strategies to Real Scenarios: Engage in practical, scenario-based knowledge checks and exercises to strengthen your ability to apply key concepts effectively.
Learning Objectives
- Describe the key benefits of taking a project management approach to GCP inspection readiness.
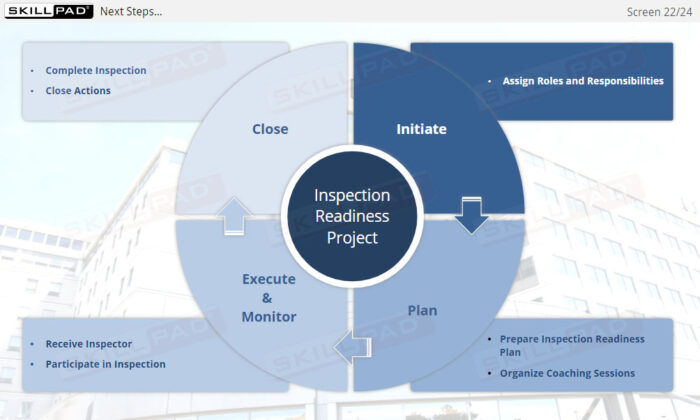
- Distinguish between the four project management phases of an inspection readiness project.
- Define the roles and responsibilities of key team members in an inspection readiness project.
- Describe the key tasks involved in the Initiate phase of an inspection readiness project.
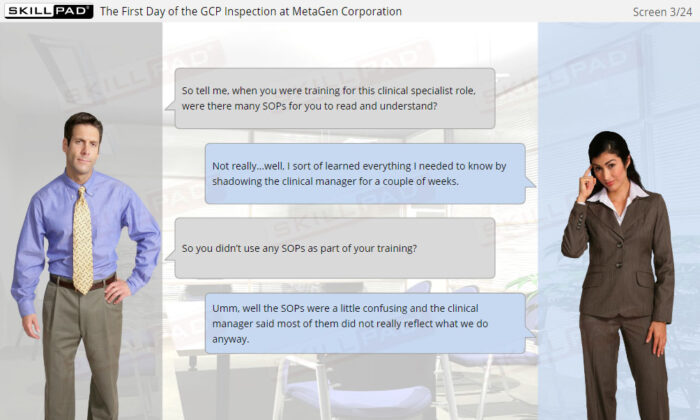
- Explain why an inspection notification must be validated.
- Describe the common project management issues associated with the Initiate phase.
- Explain how these issues can be avoided.
- Apply project management principles and techniques to the Initiate phase of inspection readiness.
Keywords
- GCP Inspection Readiness
- Project Management
- Regulatory Compliance
- Inspection Preparation
- Risk Management
- Team Roles and Responsibilities
- Inspection Notification Validation
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen