Aseptic Processing: Concepts and Controls
Aseptic processing concepts and associated technologies and techniques used for contamination control and prevention, including procedures to minimize bioburden.
Whether for onboarding or annual refresher training, this Module is seamlessly deployable on any LMS and can be tailored to your company’s exact needs.
Duration: 30 Mins
Grade: Premium Performance
Description
- Obtain a comprehensive understanding of the critical areas of aseptic processing by covering the essentials of aseptic processing, contamination control, and bioburden minimization.
- Enhance your technical vocabulary of key aseptic terms, and your understanding of the correct behaviors you should apply in an aseptic environment to ensure a safe final product.
- Better equip yourself to comply with industry regulations by understanding the importance of contamination prevention.
Learning Objectives
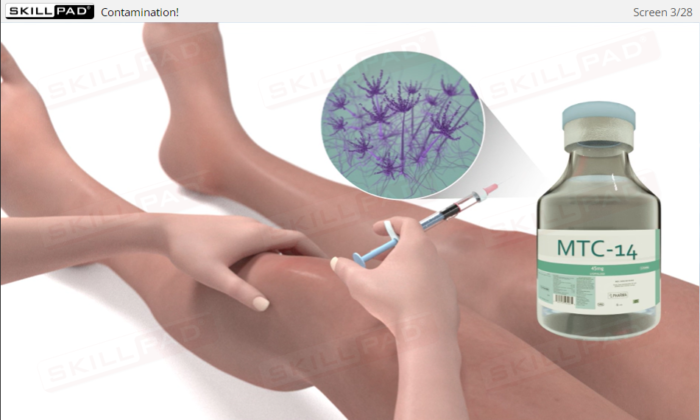
- Explain why prevention of product contamination is critical in regulated industries.
- Define the terms ‘aseptic processing’ and aseptic techniques’.
- Define the term ‘bioburden’.
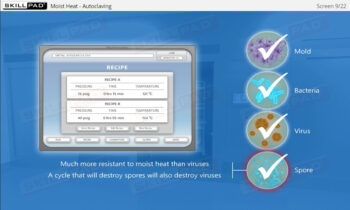
- Distinguish between the terms ‘aseptic’ and ‘sterile’.
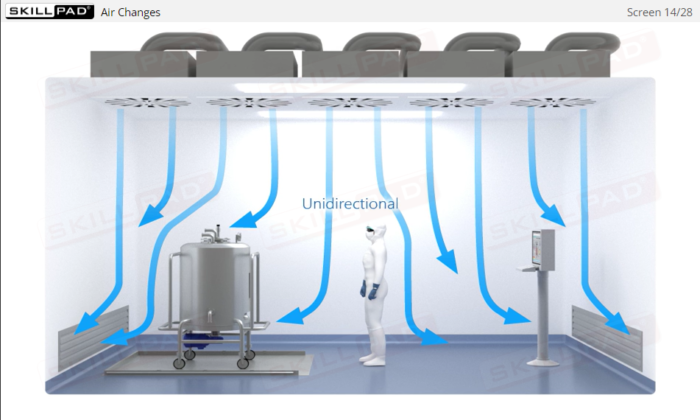
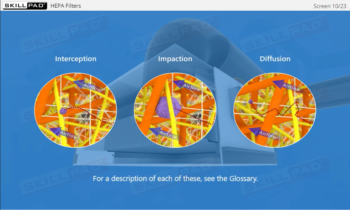
- Describe how the manufacturing environment is controlled and monitored in aseptic processing.
- Explain the role of personnel involved in aseptic processing in contributing to a safe end product.
- Apply correct behaviors when operating in an aseptic environment.
Keywords
- Aseptic Processing
- Contamination Control
- Bioburden Minimization
- Sterilization
- Cleanrooms
- Gowning
- Microbiological Monitoring
- Non-Viable Particle Monitoring
- Regulatory Compliance
- Quality Assurance.
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen