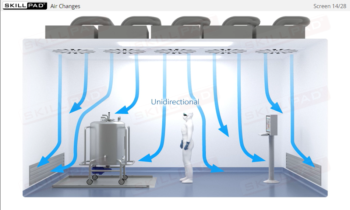
Aseptic Processing: Decontamination and Sterilization Technologies
Methods typically used to decontaminate and sterilize equipment, consumables, containers/closures, and products as part of aseptic processing. Methods covered include moist heat, dry heat, hydrogen peroxide vapor, and filtration.
Part of Annex 1 training requirements.
Whether for onboarding or annual refresher training, this Module is seamlessly deployable on any LMS and can be tailored to your company’s exact needs.
Duration: 30 Mins
Grade: Premium Performance
Description
- Gain an understanding of aseptic processing including the methods used to decontaminate and sterilize equipment, consumables, and containers/closures.
- Discover how various sterilization techniques including moist heat, dry heat, hydrogen peroxide vapor, and filtration are used.
- Recognize the importance of sterilization in aseptic processing, the process of various sterilization methods, and the concept of ‘hold times’ in relation to equipment used in aseptic processing.
Learning Objectives
- Explain why injectable products must be sterile.
- Explain the purpose of cleaning, decontamination, and sterilization in aseptic processing.
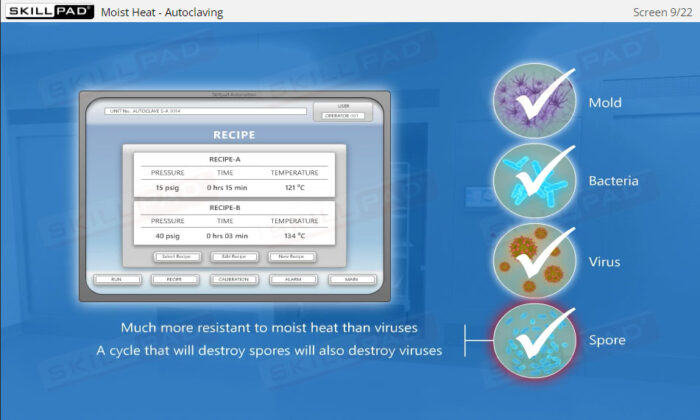
- Describe how moist heat sterilization is performed via autoclaving.
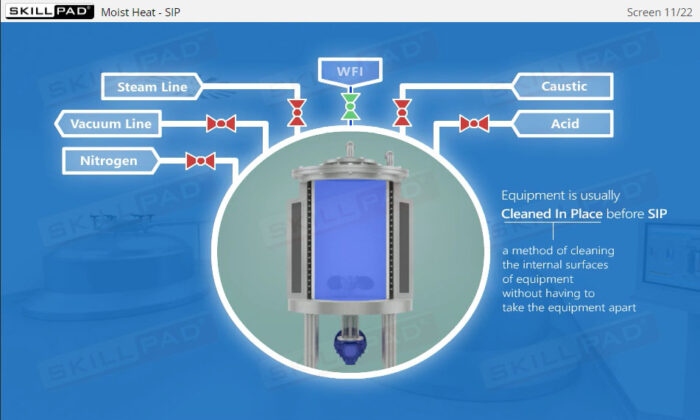
- Describe how moist heat sterilization is performed via Steam In Place.
- Describe the purpose of dry heat sterilization and how it is performed.
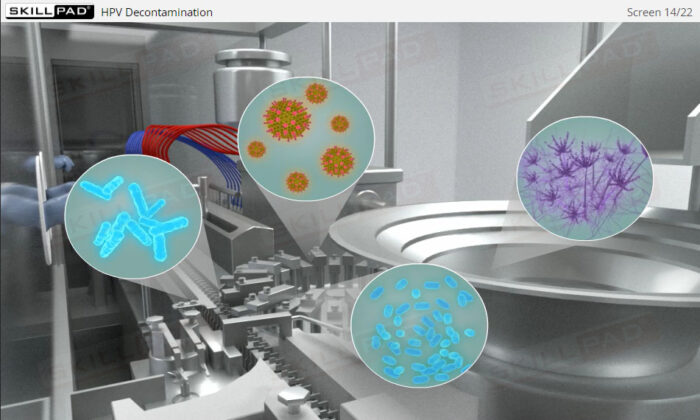
- Explain the process of HPV decontamination.
- Explain the difference between bioburden filtration and viral filtration.
- Explain what is meant by ‘hold times’ in relation to equipment used in aseptic. processing
Keywords
- Aseptic Processing
- Autoclaving
- Decontamination
- Dry Heat Sterilization
- Filtration
- Hold Times
- Hydrogen Peroxide Vapor (HPV)
- Steam In Place (SIP)
- Sterilization Technologies
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen