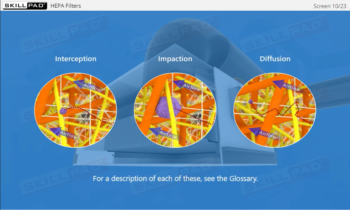
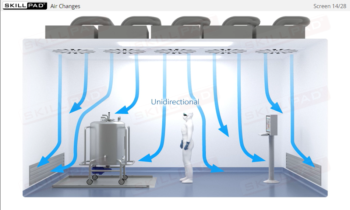
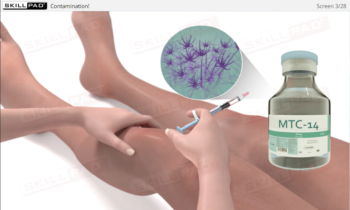
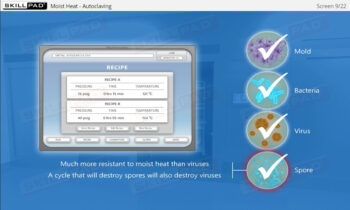
Aseptic Processing: Gowning
Gowning and its use in aseptic processing, including the different gowning requirements for different cleanroom classifications, the protective equipment used, and the procedures that are typically followed.
Part of Annex 1 training requirements.
Whether for onboarding or annual refresher training, this Module is seamlessly deployable on any LMS and can be tailored to your company’s exact needs.
Duration: 30 Mins
Grade: Premium Performance
Description
- Develop a comprehensive understanding of gowning and its role in aseptic processing, including the different gowning requirements for various cleanroom classifications.
- Demonstrate practical knowledge about the protective equipment used and the procedures typically followed in aseptic processing.
- Discover the steps required to help prevent contamination which ensures a safe final product and compliance with industry regulations.
Learning Objectives
- Define the term ‘bioburden’.
- Explain why cleanrooms are classified.
- List the four ISO cleanroom classes commonly used in Aseptic Processing.
- Explain why different cleanroom classes have different gowning requirements.
- Provide examples of gowning equipment and procedures used for different cleanroom classes.
- Explain how airlocks prevent contamination of a cleanroom environment.
Keywords
- Airlocks
- Aseptic Processing
- Bioburden
- Cleanroom Classifications
- Comprehensive Understanding
- Contamination Prevention
- Gowning
- PPE
Module Features
Animations
Voice Over
Knowledge Checks
Assessments
SCORM/AICC compatible
Full Screen